About us
A dynamic collaboration of women, scientists, engineers, physicians and clinical researchers

A message from Dr Joe Peterson, our CEO
“In the U.S., based upon insurance claims data, fewer than 40% of women have an annual mammogram beginning from the age of forty, even though ‘every year beginning at age forty,’ remains the most effective regimen and is endorsed by leaders in every clinical specialty that deals with breast cancer.
“At Bexa, our team, advisors, and board of directors are squarely focused on the lack of both participation in and access to current early detection options, a quiet but very real tragedy.
“Globally 90% of women have no technology-assisted early detection option: that’s 1.3 billion women with little chance to beat the most common women’s cancer.”

Why Bexa?
We are democratizing early detection of breast cancer because for too long we’ve seen women suffer and die from what should be, for most women, a survivable disease.
Our company’s financial structure backs up our commitment – with a model designed to accept payment from markets that can afford reasonable exam fees, and fulfill our social mission in markets that can’t.
The Bexa team includes leading physicians in breast cancer imaging, surgery and medical care. Our subject matter experts bring deep experience from some of the leading breast cancer care organizations.
We are equipped, and committed to, restoring and repairing the trust that a tone-deaf system dismantled. You can expect us to show up with full transparency, investment in our mission, and consistent action.
‘Underserved,’ in breast cancer early detection includes the majority of women of every race, social, educational and economic group. Women who choose to avoid current options. Women who can neither access nor afford expensive technology and procedures.
We choose to focus upon, and bring our highly adopted solution into populations who today are left behind.
Our corporate structure
Sure, Inc., (“Sure”) is a physician-led company that develops, owns all intellectual property related to, and manufactures the Bexa device, and operates from bases in Los Angeles, New York, Dubai (UAE), Amman (Jordan) and Colombia.
Sure provides training in the use of the Bexa device, owns and operates the SureView system, and operates continuous R&D activity.
Our history
Bexa is the result of years of innovation across multiple disciplines.
1997
Dr. Jae Son develops his new capacitive tactile pressure sensor in the Harvard Robotics Lab and forms Pressure Profile Systems, Inc. (PPS)
1999
Researchers from multiple organizations and PPS pioneer the field of pressure elastography
2001
PPS and Dr. Son form Medical Tactile, Inc. and acquire Assurance Medical’s “BreastView”, the first concept of a pressure elastography device
2003
BreastView receives FDA 510(k) clearance for creating elastographic images of abnormal breast tissues
2006
Dr. Son and MTI replace the BreastView sensors with their proprietary shape-conforming, ultra sensitive array sensor, world-leading signal conditioning electronics and add the first algorithms converting capacitive sensor data into images
2007
MTI releases this new device, “SureTouch” to the market
2013
SureTouch internal technology has evolved through continuous upgrades and performed more than 200,000 examinations in the USA and worldwide, including China, Italy, South Africa, and Australia
2015
MTI and Qualcomm collaborate to create SureTouch Mobile, a smaller, more portable device with sensor arrays connecting wirelessly to a tablet computer
MTI completes first working prototype of secure, cloud-based system for centralizing data collection and review
2016
SureTouch wins the Patrick Soon Shiong Innovation Award
2017
Sure, Inc. (“Sure,” Joe Peterson, MD, CEO) and investors acquire all SureTouch devices, technologies, rights, and intellectual property from MTI; David Ables of MTI becomes Sure’s CTO
SureTouch wins an ABBY award for innovation in healthcare delivery
2018
Sure updates clinical protocols, training programs, and market strategy
Geoff Bricker joins Sure from SOC Telemed (NASDAQ: TLMD) to lead IT
2019
SureView, the cloud-based operating ecosystem within which all devices operate and share data, is launched
Sure receives a new FDA 510K for the wireless version of SureTouch2019
SureTouch receives regulatory clearance in Saudi Arabia from FDA-equivalent, MDMA
SureTouch receives regulatory clearances in Ecuador & Jordan
TSM Technologies in Indonesia receives Qualcomm’s endorsement as contract manufacturer of SureTouch sensor units
University Cancer Center Clinical Validation Study returns landmark results
2020
SureTouch receives regulatory clearance in Mexico from FDA-equivalent, COFEPRIS
Sunny Gupta, CEO of Tachyon Systems leads the creation of machine learning and AI capacity for elastographic data into an evolving analysis
SureLearn, an online learning system supporting both remote and in-person didactic and practical training, is launched
Radiologists & primary care physicians begin investing in Sure
Quantify Health and Quantify Care become key commercialization partners in the US
2021
Robin Roberts of Novartis and COO of the Novartis Biome, joins the Sure Board of Directors
SURE begins a large-scale definitive clinical validation study with a leading US academic imaging department slated for completion in July of 2021
Intelense adapts their industry-leading automated video analysis and anomaly detection engine into new analysis and visualization algorithms for elastography data
The SureView AI system expands to include the ability to “dream”
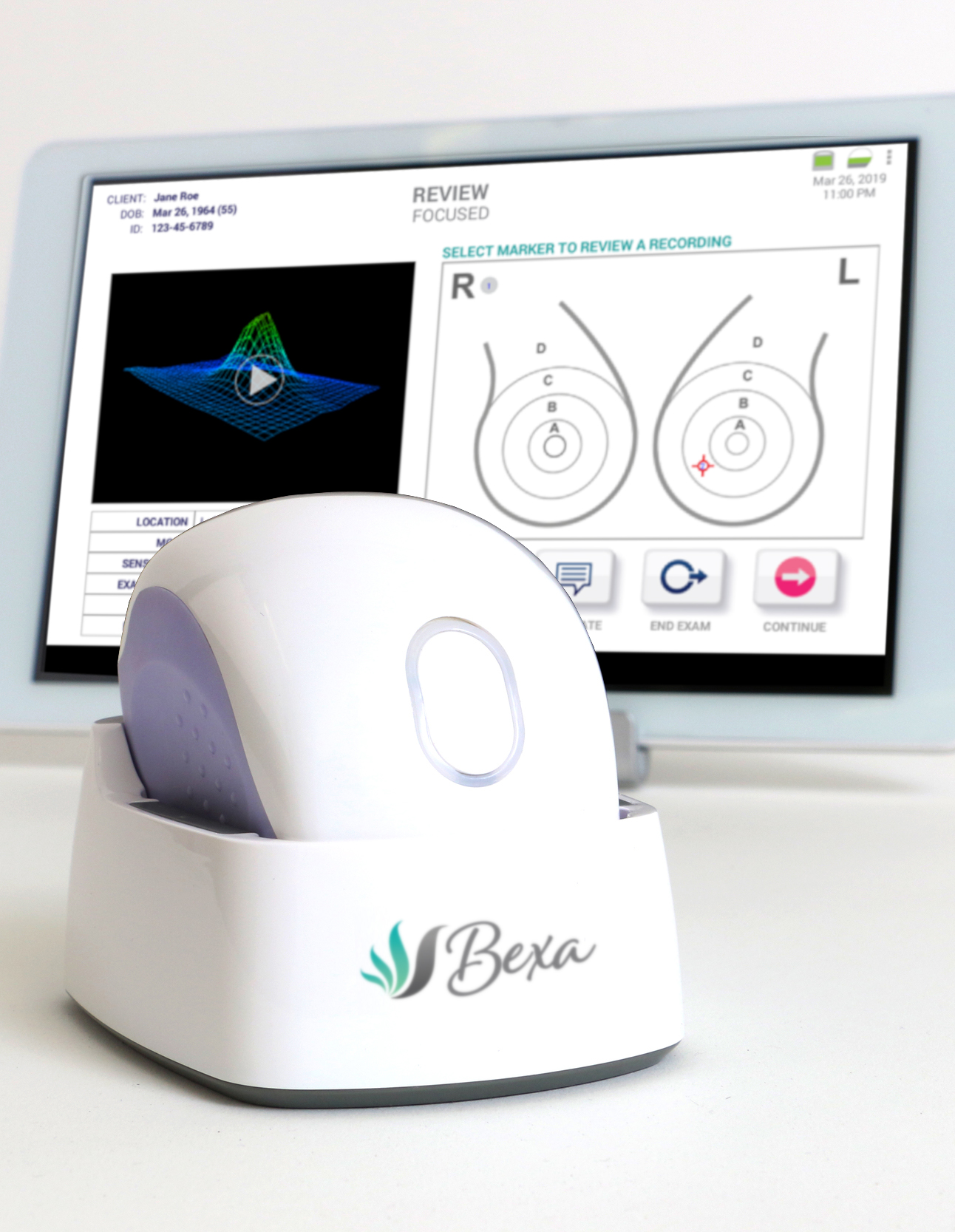
The transformed SureTouch is re-released as Bexa

Our team
At Bexa, our mission is ‘no woman left behind’
Our team is a dynamic collaboration of women, scientists, engineers, physicians, and clinical researchers.
Thank you for taking the time to learn about Bexa
We’re working hard to continuously advance the sophistication of Bexa and to establish its role in the early detection of breast cancer.